Dr Baptiste BALANCA
Service de neuroanesthésie et réanimation à l’hôpital Pierre Wertheimer
The neurological hospital’s anesthesia-resuscitation team intervenes during the 4,500 procedures carried out each year by the establishment’s surgeons and radiologists. It supports more than 1,600 intensive care or post-interventional care stays/year. Specialized, it also provides plasmapheresis treatments for patients suffering from chronic dysimmune pathologies.
This is a highly specialized team due to its regional recruitment for one of the largest neurological specialty centers in France. Ensuring them are taken care of:
- vascular emergencies at the neurological hospital for patients in vital distress
- critical care after the heaviest surgeries and/or for the most fragile patients
- anesthesia for highly specialized surgeries
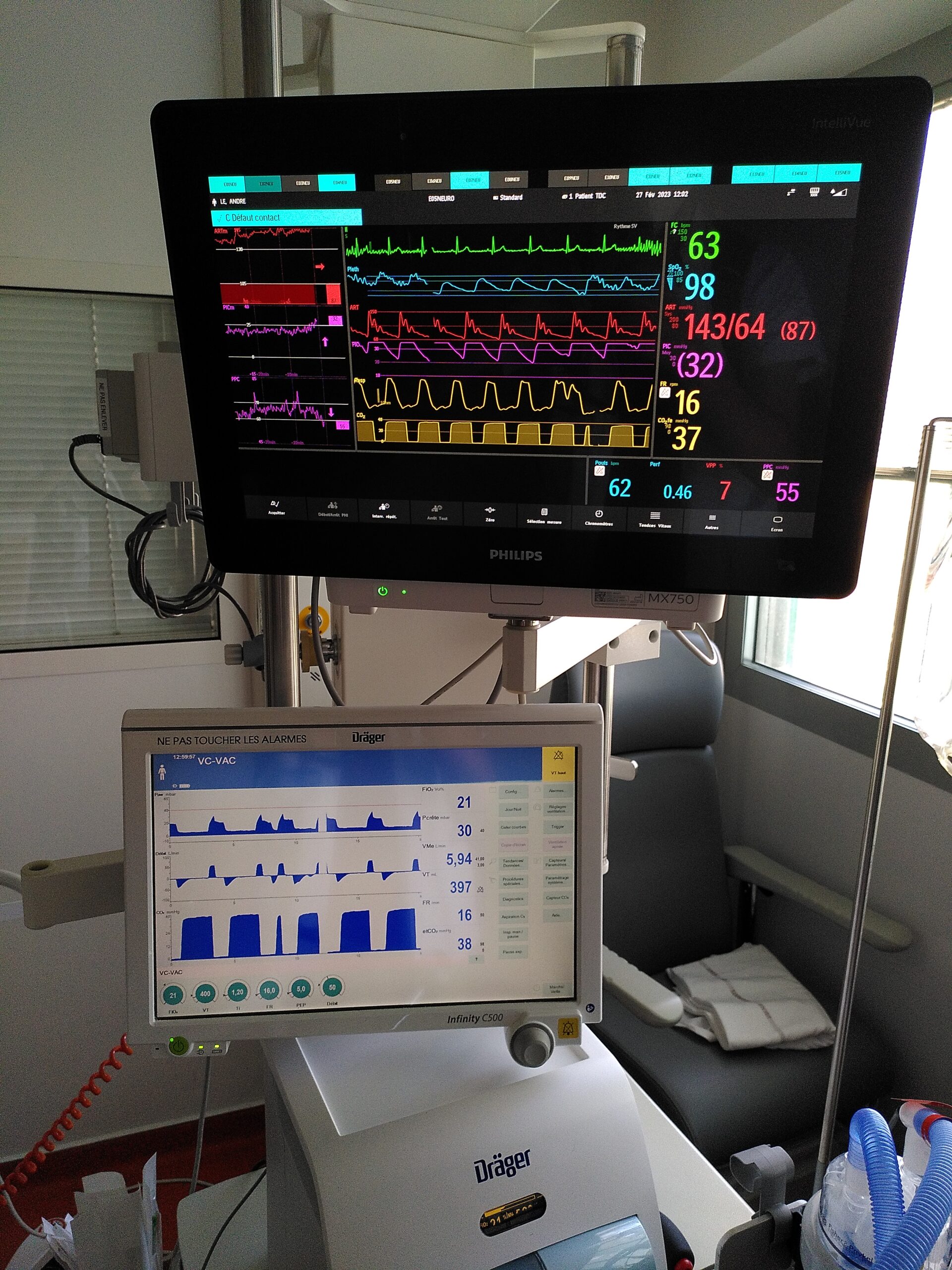
The teams are equipped with the most modern technologies and constantly updated therapies to guarantee these missions. In particular, innovative anesthesia techniques are being developed for awake interventions on brain lesions in the critical functional area of language or motor skills, which reduces the after-effects of these interventions.

Equipe de Recherche: CRNL – TIGER
Theme: Severe Acute Brain Injuries
The progression of cerebral lesions after an acute brain injury (traumatic, ischemic or hemorrhagic) is still poorly understood. Detection and prevention are therefore difficult in patients hospitalized in the intensive care unit (ICU) with consciousness disorders that compromise neurological examination, with the result that up to a third of them have lesions discovered on follow-up imaging. Our aim is to understand the pathophysiology of the progression of these brain lesions, in order to identify new markers and therapeutic targets to improve and individualize management strategies. Our team is conducting translational research into several aspects of brain function during acute brain injuries, using data from multimodal monitoring of intensive care patients and preclinical models in rodents and non-human primates:
– Role of cortical spreading depolarizations after subarachnoid hemorrhage or traumatic brain injury.
– Cerebral vascularization, metabolism and autoregulation after brain injury, and the impact of anesthetic agents.
– EEG rhythm changes, and studies of vegetative functions from the acute phase through to neurological recovery.
Publications
1. Early brain metabolic disturbances associated with delayed cerebral ischemia in patients with severe subarachnoid hemorrhage
Tholance Y, Aboudhiaf S, Balança B, Barcelos GK, Grousson S, Carrillon R, Lieutaud T, Perret-Liaudet A, Dailler F, Marinesco S
J Cereb Blood Flow Meta (2023) — Abstract
Delayed cerebral ischemia (DCI) is a devastating complication of aneurysmal subarachnoid hemorrhage (ASAH) causing brain infarction and disability. Cerebral microdialysis (CMD) monitoring is a focal technique that may detect DCI-related neurochemical changes as an advance warning. We conducted retrospective analyses from 44 poor-grade ASAH patients and analyzed glucose, lactate, pyruvate, and glutamate concentrations in control patients without DCI (n = 19), and in patients with DCI whose CMD probe was located within (n = 17) or outside (n = 8) a new infarct. When monitored from within a lesion, DCI was preceded by a decrease in glucose and a surge in glutamate, accompanied by increases in lactate/pyruvate and lactate/glucose ratios whereas these parameters remained stable in control patients. When CMD monitoring was performed outside the lesion, the glutamate surge was absent, but glucose and L/G ratio were still significantly altered. Overall, glucose and L/G ratio were significant biomarkers of DCI (se96.0, spe73.7-68.4). Glucose and L/G predicted DCI 67 h before CT detection of a new infarct. The pathogenesis of DCI therefore induces early metabolic disturbances that can be detected by CMD as an advance warning. Glucose and L/G could provide a trigger for initiating further examination or therapy, earlier than when guided by other monitoring techniques.
Keywords: Brain metabolism; cerebral microdialysis; glucose; lactate; multimodal monitoring.
2. Outcomes After Endovascular Therapy With Procedural Sedation vs General Anesthesia in Patients With Acute Ischemic Stroke: The AMETIS Randomized Clinical Trial
Chabanne R, Geeraerts T, Begard M, Balança B, Rapido F, Degos V, Tavernier B, Molliex S, Velly L, Verdonk F, Lukaszewicz AC, Perrigault PF, Albucher JF, Cognard C, Guyot A, Fernandez C, Masgrau A, Moreno R, Ferrier A, Jaber S, Bazin JE, Pereira B, Futier E; ANARLF NetworkAMETIS Study Group
JAMA Neurol. (2023)— Abstract
Importance: General anesthesia and procedural sedation are common practice for mechanical thrombectomy in acute ischemic stroke. However, risks and benefits of each strategy are unclear.
Objective: To determine whether general anesthesia or procedural sedation for anterior circulation large-vessel occlusion acute ischemic stroke thrombectomy are associated with a difference in periprocedural complications and 3-month functional outcome.
Design, setting, and participants: This open-label, blinded end point randomized clinical trial was conducted between August 2017 and February 2020, with final follow-up in May 2020, at 10 centers in France. Adults with occlusion of the intracranial internal carotid artery and/or the proximal middle cerebral artery treated with thrombectomy were enrolled.
Interventions: Patients were assigned to receive general anesthesia with tracheal intubation (n = 135) or procedural sedation (n = 138).
Main outcomes and measures: The prespecified primary composite outcome was functional independence (a score of 0 to 2 on the modified Rankin Scale, which ranges from 0 [no neurologic disability] to 6 [death]) at 90 days and absence of major periprocedural complications (procedure-related serious adverse events, pneumonia, myocardial infarction, cardiogenic acute pulmonary edema, or malignant stroke) at 7 days.
Results: Among 273 patients evaluable for the primary outcome in the modified intention-to-treat population, 142 (52.0%) were women, and the mean (SD) age was 71.6 (13.8) years. The primary outcome occurred in 38 of 135 patients (28.2%) assigned to general anesthesia and in 50 of 138 patients (36.2%) assigned to procedural sedation (absolute difference, 8.1 percentage points; 95% CI, -2.3 to 19.1; P = .15). At 90 days, the rate of patients achieving functional independence was 33.3% (45 of 135) with general anesthesia and 39.1% (54 of 138) with procedural sedation (relative risk, 1.18; 95% CI, 0.86-1.61; P = .32). The rate of patients without major periprocedural complications at 7 days was 65.9% (89 of 135) with general anesthesia and 67.4% (93 of 138) with procedural sedation (relative risk, 1.02; 95% CI, 0.86-1.21; P = .80).
Conclusions and relevance: In patients treated with mechanical thrombectomy for anterior circulation acute ischemic stroke, general anesthesia and procedural sedation were associated with similar rates of functional independence and major periprocedural complications.
Trial registration: ClinicalTrials.gov Identifier: NCT03229148.
3. Effect of an individualized versus standard blood pressure management during mechanical thrombectomy for anterior ischemic stroke: the DETERMINE randomized controlled trial
Maïer B, Gory B, Chabanne R, Tavernier B, Balanca B, Audibert G, Thion LA, Le Guen M, Geeraerts T, Calviere L, Degos V, Lapergue B, Richard S, Djarallah A, Mophawe O, Boursin P, Le Cossec C, Blanc R, Piotin M, Mazighi M, Gayat E; DETERMINE Investigators
Trials. (2022) — Abstract
Background: Hypotension and blood pressure (BP) variability during endovascular therapy (EVT) for acute ischemic stroke (AIS) due to an anterior large vessel occlusion (LVO) is associated with worse outcomes. However, the optimal BP threshold during EVT is still unknown given the lack of randomized controlled evidence. We designed the DETERMINE trial to assess whether an individualized BP management during EVT could achieve better functional outcomes compared to a standard BP management.
Methods: The DETERMINE trial is a multicenter, prospective, randomized, controlled, open-label, blinded endpoint clinical trial (PROBE design). AIS patients with a proximal anterior LVO are randomly assigned, in a 1:1 ratio, to an experimental arm in which mean arterial pressure (MAP) is maintained within 10% of the first MAP measured before EVT, or a control arm in which systolic BP (SBP) is maintained within 140-180 mm Hg until reperfusion is achieved or artery closure in case of EVT failure. The primary outcome is the rate of favorable functional outcomes, defined by a modified Rankin Scale (mRS) between 0 and 2 at 90 days. Secondary outcomes include excellent outcome and ordinal analysis of the mRS at 90 days, early neurological improvement at 24 h (National Institutes of Health Stroke Scale), final infarct volume, symptomatic intracranial hemorrhage rates, and all-cause mortality at 90 days. Overall, 432 patients will be included.
Discussion: DETERMINE will assess the clinical relevance of an individualized BP management before reperfusion compared to the one size fits all approach currently recommended by international guidelines.
Trial registration: ClinicalTrials.gov , NCT04352296.
4. Cortical Spreading Depolarizations in Aneurysmal Subarachnoid Hemorrhage: An Overview of Current Knowledge and Future Perspectives
Berhouma M, Eker OF, Dailler F, Rheims S, Balanca B
Adv Tech Stand Neurosurg. (2022) — Abstract
Despite significant advances in the management of aneurysmal subarachnoid hemorrhage (SAH), morbidity and mortality remain devastating particularly for high-grade SAH. Poor functional outcome usually results from delayed cerebral ischemia (DCI). The pathogenesis of DCI during aneurysmal SAH has historically been attributed to cerebral vasospasm, but spreading depolarizations (SDs) are now considered to play a central role in DCI. During SAH, SDs may produce an inverse hemodynamic response leading to spreading ischemia. Several animal models have contributed to a better understanding of the pathogenesis of SDs during aneurysmal SAH and provided new therapeutic approaches including N-methyl-D-aspartate receptor antagonists and phosphodiesterase inhibitors. Herein we review the current knowledge in the field of SDs’ pathogenesis and we detail the key experimental and clinical studies that have opened interesting new therapeutic approaches to prevent DCI in aneurysmal SAH.
Keywords: Cerebral aneurysm; Delayed cerebral ischemia; Microcirculation; Spreading depolarization; Spreading ischemia; Subarachnoid hemorrhage.
5. How to monitor thiopental administration in the intensive care unit for refectory status epilepticus or intracranial hypertension?
Dabricot E, Seqat I, Dailler F, Rheims S, Boulogne S, Balança B
Crit Care. (2021) — Abstract
6. DAMPs and RAGE Pathophysiology at the Acute Phase of Brain Injury: An Overview
Balança B, Desmurs L, Grelier J, Perret-Liaudet A, Lukaszewicz AC
Int J Mol Sci. (2021) — Abstract
Early or primary injury due to brain aggression, such as mechanical trauma, hemorrhage or is-chemia, triggers the release of damage-associated molecular patterns (DAMPs) in the extracellular space. Some DAMPs, such as S100B, participate in the regulation of cell growth and survival but may also trigger cellular damage as their concentration increases in the extracellular space. When DAMPs bind to pattern-recognition receptors, such as the receptor of advanced glycation end-products (RAGE), they lead to non-infectious inflammation that will contribute to necrotic cell clearance but may also worsen brain injury. In this narrative review, we describe the role and ki-netics of DAMPs and RAGE at the acute phase of brain injury. We searched the MEDLINE database for “DAMPs” or “RAGE” or “S100B” and “traumatic brain injury” or “subarachnoid hemorrhage” or “stroke”. We selected original articles reporting data on acute brain injury pathophysiology, from which we describe DAMPs release and clearance upon acute brain injury, and the implication of RAGE in the development of brain injury. We will also discuss the clinical strategies that emerge from this overview in terms of biomarkers and therapeutic perspectives.
Keywords: acute brain injuries; biomarkers; damage-associated molecular pattern molecules; receptor for advanced glycation end-products.
7. Significance and Diagnostic Accuracy of Early S100B Serum Concentration after Aneurysmal Subarachnoid Hemorrhage
Balança B, Ritzenthaler T, Gobert F, Richet C, Bodonian C, Carrillon R, Terrier A, Desmurs L, Perret-Liaudet A, Dailler F
J Clin Med. (2020) — Abstract
Background: Early brain injuries (EBI) are one of the most important causes of morbidity and mortality after subarachnoid hemorrhage. At admission, a third of patients are unconscious (spontaneously or sedated) and EBI consequences are not evaluable. To date, it is unclear who will still be comatose (with severe EBI) and who will recover (with less severe EBI) once the aneurysm is treated and sedation withdrawn. The objective of the present study was to determine the diagnostic accuracy of S100B levels at hospital admission to identify patients with severe neurological consequences of EBI.
Methods: Patients were consecutively included in this prospective blinded observational study. A motor component of the Glasgow coma score under 6 on day 3 was used to define patients with severe neurological consequences of EBI.
Results: A total of 81 patients were included: 25 patients were unconscious at admission, 68 were treated by coiling. On day 3, 12 patients had severe consequences of EBI. A maximal S100B value between admission and day 1 had an area under the receiver operating characteristic curve (AUC) of 86.7% to predict severe EBI consequences. In patients with impaired consciousness at admission, the AUC was 88.2%.
Conclusion: Early S100B seems to have a good diagnostic value to predict severe EBI. Before claiming the usefulness of S100B as a surrogate marker of EBI severity to start earlier multimodal monitoring, these results must be confirmed in an independent validation cohort.
Keywords: S100B protein; early brain injury; patient outcome assessment; subarachnoid hemorrhage.
8. Diagnostic accuracy of quantitative EEG to detect delayed cerebral ischemia after subarachnoid hemorrhage: A preliminary study
Balança B, Dailler F, Boulogne S, Ritzenthaler T, Gobert F, Rheims S, Andre-Obadia N
Clin Neurophysiol. (2018) — Abstract
Objective: Delayed cerebral ischemia (DCI) is the most important and preventable morbidity cause after subarachnoid hemorrhage (SAH). Therefore, DCI early detection is a major challenge. Yet, neurological examination can be unreliable in poor grade SAH patients. EEG provides information from most superficial cortical area, with ischemia-related changes. This study aims at defining an alpha-theta/delta (AT/D) ratio decrease thresholds to detect DCI.
Methods: We used EEG with a montage matching vascular territories (right and left anterior central and posterior) and compared them to follow-up brain imaging.
Results: 15 SAH patients (Fischer ≥ 3, World Federation of Neurological Surgeons scale ≥4, 9 DCI) were monitored during 6.4 [4-8] days (min = 2d, max = 13d). AT/D changes could follow three different patterns: (1) prolonged or (2) transient decrease and (3) no decrease or progressive increase. A regional 30% decrease outlasting 3.7 h reached 100% sensitivity and 88.9% specificity to detect DCI. Only 22.6% were in a zone of uncertain diagnosis (3.7-8.04 h). These prolonged decreases, with a loss of transient changes, started in cortical areas evolving toward DCI, and preceded intracranial changes when available.
Conclusion: Although this study has a small sample size, prolonged AT/D decrease seems to be a reliable biomarker of DCI.
Significance: cEEG changes are likely to precede cerebral infarction and could be useful at the bedside to detect DCI before irreversible damage.
Keywords: Alpha-theta/delta ratio; Continuous electroencephalography; Delayed cerebral ischemia; Sub-arachnoid hemorrhage.
9. Altered hypermetabolic response to cortical spreading depolarizations after traumatic brain injury in rats
Balança B, Meiller A, Bezin L, Dreier JP, Marinesco S, Lieutaud T
J Cereb Blood Flow Metab. (2017) — Abstract
Spreading depolarizations are waves of near-complete breakdown of neuronal transmembrane ion gradients, free energy starving, and mass depolarization. Spreading depolarizations in electrically inactive tissue are associated with poor outcome in patients with traumatic brain injury. Here, we studied changes in regional cerebral blood flow and brain oxygen (PbtO2), glucose ([Glc]b), and lactate ([Lac]b) concentrations in rats, using minimally invasive real-time sensors. Rats underwent either spreading depolarizations chemically triggered by KCl in naïve cortex in absence of traumatic brain injury or spontaneous spreading depolarizations in the traumatic penumbra after traumatic brain injury, or a cluster of spreading depolarizations triggered chemically by KCl in a remote window from which spreading depolarizations invaded penumbral tissue. Spreading depolarizations in noninjured cortex induced a hypermetabolic response characterized by a decline in [Glc]b and monophasic increases in regional cerebral blood flow, PbtO2, and [Lac]b, indicating transient hyperglycolysis. Following traumatic brain injury, spontaneous spreading depolarizations occurred, causing further decline in [Glc]b and reducing the increase in regional cerebral blood flow and biphasic responses of PbtO2 and [Lac]b, followed by prolonged decline. Recovery of PbtO2 and [Lac]b was significantly delayed in traumatized animals. Prespreading depolarization [Glc]b levels determined the metabolic response to clusters. The results suggest a compromised hypermetabolic response to spreading depolarizations and slower return to physiological conditions following traumatic brain injury-induced spreading depolarizations.
Keywords: Biosensors; brain metabolism; brain trauma; cerebral blood flow; energy metabolism; spreading depolarization; spreading depression.
10. Recording, analysis, and interpretation of spreading depolarizations in neurointensive care: Review and recommendations of the COSBID research group
Dreier JP and al.
J Cereb Blood Flow Metab. (2017) — Abstract
Spreading depolarizations (SD) are waves of abrupt, near-complete breakdown of neuronal transmembrane ion gradients, are the largest possible pathophysiologic disruption of viable cerebral gray matter, and are a crucial mechanism of lesion development. Spreading depolarizations are increasingly recorded during multimodal neuromonitoring in neurocritical care as a causal biomarker providing a diagnostic summary measure of metabolic failure and excitotoxic injury. Focal ischemia causes spreading depolarization within minutes. Further spreading depolarizations arise for hours to days due to energy supply-demand mismatch in viable tissue. Spreading depolarizations exacerbate neuronal injury through prolonged ionic breakdown and spreading depolarization-related hypoperfusion (spreading ischemia). Local duration of the depolarization indicates local tissue energy status and risk of injury. Regional electrocorticographic monitoring affords even remote detection of injury because spreading depolarizations propagate widely from ischemic or metabolically stressed zones; characteristic patterns, including temporal clusters of spreading depolarizations and persistent depression of spontaneous cortical activity, can be recognized and quantified. Here, we describe the experimental basis for interpreting these patterns and illustrate their translation to human disease. We further provide consensus recommendations for electrocorticographic methods to record, classify, and score spreading depolarizations and associated spreading depressions. These methods offer distinct advantages over other neuromonitoring modalities and allow for future refinement through less invasive and more automated approaches.
Keywords: Spreading depolarization; anoxic depolarization; asphyxial depolarization; brain edema; brain trauma; cerebral blood flow; epilepsy; epileptogenesis; focal ischemia; global ischemia; intracerebral hemorrhage; neurocritical care; neuroprotection; neurovascular coupling; peri-infarct depolarization; spreading depression; spreading ischemia; subarachnoid hemorrhage; vasospasm.

